Our team aims to decipher how ApoE4 drives tau-mediated synaptic degeneration in AD by 1) identifying at tau-damaged synapses the ApoE4-mediated functional and structural synaptic changes; 2) identifying the protein players underlying these structural and functional changes as well as their cellular origin; 3) designing treatments targeting ApoE or its downstream effectors to promote synaptic welfare in AD. The rationale behind is that targeting synaptic dysfunctions becomes an attractive therapeutic strategy because they occur at early stages of the disease and because preserving synapses is crucial for brain function and cognition. Yet counteracting synaptic loss has been relatively neglected up to date because synaptotoxic mechanisms are still poorly known (scientific challenge), and because of the difficulty in reaching synaptic resolution (technological challenge). My team aims to address the scientific challenge by investigating the role of the strongest genetic risk factor in AD (ApoE4) on tau-mediated synaptic degeneration, taking into account the role of glial cells. We also aim to overcome the technological roadblock by achieving a high-level experimental synaptic resolution using the synergistic power of advanced proteomics, chemogenetics and super resolution microscopy at the synapse level, combined with electrophysiology and behavioral testing.
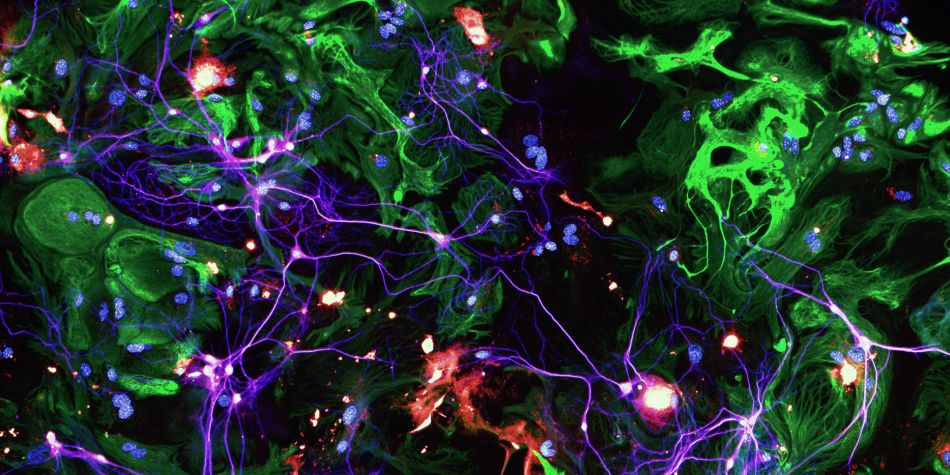
Alzheimer’s disease (AD) is one of the biggest societal and economical challenges of the 21st century for which no cure exists at this point. AD is characterized by the accumulation of amyloid-β (Aβ) in plaques and the aggregation of tau protein in neurons (tau pathology). In AD, the progressive accumulation of tau pathology leads to the degeneration of synapses (contact zone between two neurons where the transmission of the nervous message takes place), and, consequently, the well-known memory disorders in AD.
Apolipoprotein E (ApoE) is a protein essential for synaptic functions. The E4 isoform of ApoE (ApoE4) strongly increases the risk of developing AD. Although our previous studies show that ApoE4 exacerbates synaptic loss, we don't know how. My work aims to characterize with a synaptic resolution, how ApoE4 controls tau-mediated synaptic degeneration in AD, and to use this new knowledge to design effective therapeutic strategies against this incurable disease.
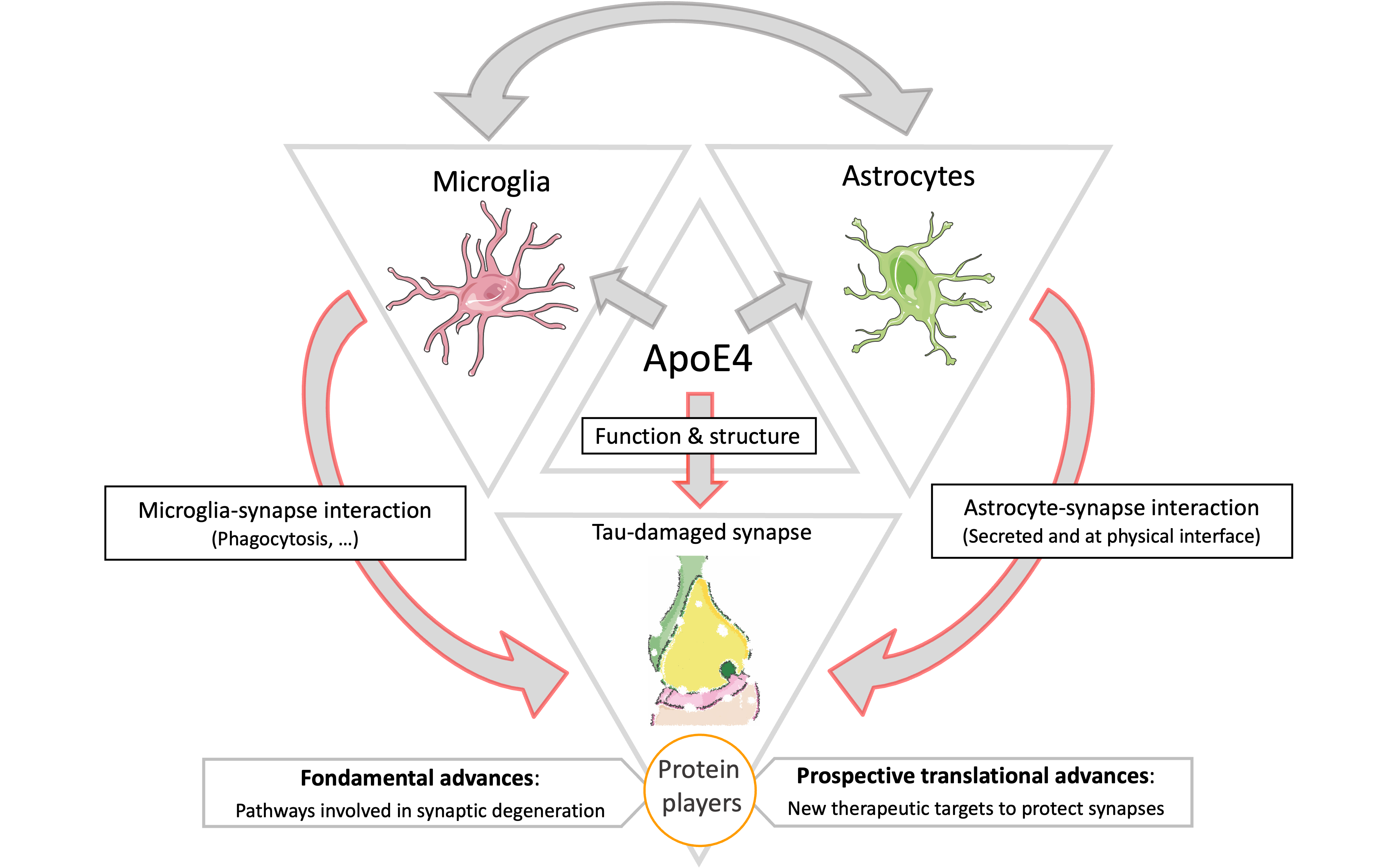
For this, I have developed 3 axes of research:
1) Synaptic changes: A first research axis of the team aims to identify how ApoE4 modifies the functions and structures of tau-damaged synapses.
2) Cellular and protein players: A second axis of research aims to determine whether ApoE4 requires microglia (immune brain cells) and/or astrocytes (providing both metabolic and physical support to neurons) to exacerbate synaptic loss in AD. In addition, we are aiming to identify the proteins responsible for synaptic loss at the synapses, as well as the cellular origin of these proteins (neurons, astrocytes or microglia).
3) Treatments: Finally, our last axis of research is to identify new therapeutic targets targeting ApoE4, but also the protein and cellular actors identified in the second research axis in order to protect synapses in AD.
These three research topics will help to better understand the relationships between microglia, astrocytes and tau pathology leading to synaptic loss, in order to find new therapeutic targets to protect synapses in Alzheimer's disease.
Determining with unprecedented synaptic resolution how ApoE4 exacerbates tau-mediated synaptic degeneration in AD requires a fine characterization of ApoE4 functions at tau-damaged synapses. This strategy is a cornerstone in validating the synapse as a target for which developing innovative strategies to promote synaptic welfare in AD.
Our team hypothesizes that ApoE4 contributes to tau-mediated synaptic degeneration by altering glia/neuron interactions.
1: SYNAPTIC CHANGES: In vivo studies have clearly shown that ApoE4 exacerbates tau-mediated pathologies (Gratuze et al., 2023; Shi et al., 2017; Chao Wang et al., 2021; Zhao et al., 2020). In a recent study, I reported that ApoE4 lowers post-synaptic PSD95 in P301S mice (Gratuze et al., 2023), suggesting that ApoE4 worsens tau-mediated synaptic degeneration. Because of ApoE’s role in synaptic functions in AD, we aim to determine how ApoE isoforms affect tau-mediated synaptic dysfunction. We aim to identify functional and structural synaptic changes caused by ApoE4 at tau-damaged synapses in the presence or not of microglia by combining super resolution microscopy, electrophysiology and behavioral tests.
2: PROTEIN PLAYERS: We previously showed that ApoE4 exacerbates synaptic alterations in P301S mice, but it remains unclear which proteins underlie this phenomenon, their cellular origin and their dependence on the molecular interactions between cell types (i.e., neurons and astrocytes). Understanding the pathogenic mechanisms behind the ApoE4-tau axis is crucial to elucidate the role of ApoE in synaptic impairments, and by extension also the role of astrocytes, since they are the main source of ApoE. Accordingly, we are assessing how ApoE isoforms affect the astrocyte- or neuron-derived nascent proteome (i.e., newly synthesized proteins) at synapses. Moreover, we are evaluating the ApoE isoform-specific changes at the interface astrocyte/synapses using an in vivo chemogenetic approach based on cell surface fragment complementation.We want to identify the protein players underlying the ApoE4-associated structural and functional changes described in the first research topic, combining chemogenetic approaches and advanced proteomics to specifically label and quantify proteins produced by neurons or astrocytes at: 1) synapses, and 2) the interface between astrocytes and synapses.
3: TREATMENT: AD is a dramatic public health problem with around 50 million people affected worldwide, highlighting the urgency of developing strategies to slow/prevent/cure AD. In this regard, it is estimated that a treatment that delays AD onset by 5 years would reduce the number of cases by 50%. Because of ApoE4’s role in both synaptic impairments and tau-mediated neurodegeneration, we propose to develop and test therapies targeting 1) ApoE and ApoE receptors, and 2) the most promising protein players at tau-damaged synapses identified in the second research topic to protect synapses in AD.



















